Human Serum & Animal-Free Growth Supplement
ZenBio is proud to offer several non-animal replacements for Fetal Bovine Serum (FBS): Human Platelet Lysate-(New!), GroPro ready-to-use growth supplement and Human AB Serum.

ZenBio, Inc. Introduces GroPro™ Cell Culture Growth Supplement Formulated to Replace Bovine Serum Allowing for Culturing of Primary Cells and Cell Lines
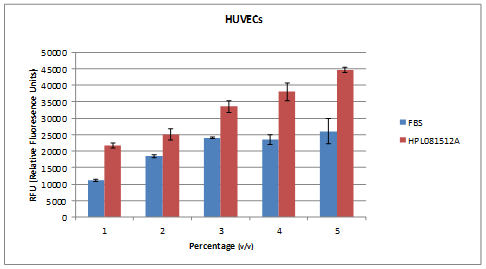
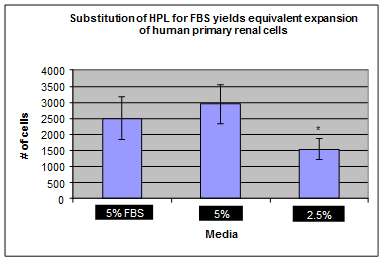
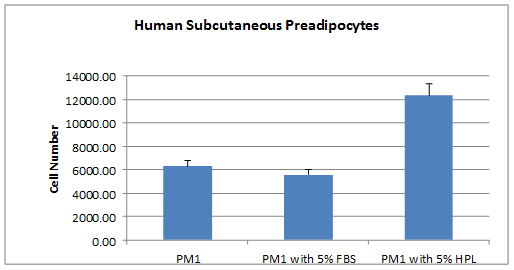
Our NEW! Human Platelet Lysate (HPL) and GroPro are a non-animal derived cell culture growth supplements obtained from human platelets. HPL contains abundant growth factors and cytokines necessary for cell growth and proliferation. Platelet lysate can be used as a replacement for traditional Fetal Bovine (FBS) supplemented cell culture medium. Replacing fetal bovine serum(FBS) with HPL allows for expansion and potential clinical grade production of functional mesenchymal stromal cells by removing the risk of xenogeneic immune reactions and transmission of bovine prion and viral pathogens. Additionally, HPL and GroPro are derived from a renewable source that is not tied to the animal slaughter industry and the price does not fluctuate!

The US FDA is moving towards adopting European guidelines requiring that bovine materials be removed from culturing media for any therapeutic product containing cultured cells, thus making HPL an attractive alternative to the R&D and production/manufacturing processes. All lots are fully tested for pathogens according to FDA guidelines.
- 10X more potent than FBS
- Stable Pricing
- Non-Animal Derived
- Traceability
- Free of Mycoplasma
- Lot-to-Lot Consistency
- Sterile Collection
- Pool Processing
- QC Tested in Zen Bio's Cell Production System
 |
 |
 |
Human AB Serum:

Human serum offers a technical advantage over animal serum when culturing a variety of human cell types, especially those cells making up the immune system. This is also the serum of choice when developing tissue engineered and regenerative medicine products, as many regulatory agencies in a growing number of countries are restricting the use of animal serum during the manufacturing process.
Since human serum from type AB donors is lacking in antibodies towards the A and B blood-type antigens, this is the type most commonly used as immunoreactivity is minimized against the human cells in culture. Male donor serum is preferred over female, as this lowers the risk of major histocompatibility class (MHC) antibodies being present. Women having a past pregnancy may have developed these antibodies to MHC antigens carried on the father's cells or the developing fetus.
Ordering Information:
| Item# | Item Desc | U/M | Price |
|---|---|---|---|
| SER-HPL | Human Platelet Lysate, Concentrated , Sterile | 100ml | $435.00 |
| SER-HPL-GROPRO | GroPro CLEAR Cell Culture Growth Supplement, Animal Component Free, Human Platelet Lysate-READY TO USE, Sterile Filtered-Frozen | 500ml | $209.00 |
| HSER-ABP100ML | Human AB Serum, Off-clot, Pooled Donor lot, Sterile Filtered | 100ml | $367.00 |
| HSER-ABPM100ML | Human AB Serum, Off-the-clot, Pooled MALE Donor lot, Sterile Filtered | 100ml | $409.00 |
| HSER-CABPM100ML | Human AB Serum, (Converted) pooled MALE ONLY donor lot, Heat Inactivated, STERILE 100ml | 100ml | $235.00 |
Call for a quote on bulk quantities.
Please contact us for pricing.
Human Serum & Animal-Free Growth Supplement Publications
Human periodontal ligament stem cells on calcium phosphate scaffold delivering platelet lysate to enhance bone regeneration
Zeqing Zhao, Jin Liu, Michael D. Weir, Ning Zhang, Li Zhang, Xianju Xie, Charles Zhang, Ke Zhang, Yuxing Bai and Hockin H. K. XuDOI: 10.1039/C9RA08336G (Paper) RSC Adv., 2019, 9, 41161-41172
Biocompatibility Evaluation of ProRoot MTA Mixed with Human Platelet Lysate on Human Osteoblastic Cells In Vitro Studies
Tariq AqiliCharacterization and comparison of human limbal explant cultures grown under defined and xeno-free conditions
Kristyna Brejchova, Peter Trosan, Pavel Studeny, Pavlina Skalicka, Tor Paaske Utheim, Jan Bednar, Katerina Jirsovahttps://www.doi.org/10.1016/j.exer.2018.06.019
Transcutaneously refillable, 3D-printed biopolymeric encapsulation system for the transplantation of endocrine cells
Marco Farina, Corrine Ying Xuan Chua, Andrea Ballerinia, Usha Thekkedath, Jenolyn F. Alexander, Jessica R. Rhudy, Gianluca Torchio, Daniel Fraga, Ravi R. Pathak, Mariana Villanueva, Crystal S. Shin, Jean A. Niles, Raffaella Sesana, Danilo Demarchi,https://www.doi.org/10.1016/j.biomaterials.2018.05.047
Fabrication and Characterization of NanoCalcium Sulfate and Human Platelet Lysate as a Growth Factor Delivery System
Andrew Barone, Adam Morrell, and Rosemary DziakInt J Dent Oral Health 2(4): http://dx.doi.org/10.16966/2378-7090.172
Platelet Lysate as Replacement for Fetal Bovine Serum in Limbal Stem Cell Cultures: Preliminary Results
Kunal Suri; Ching Yuan; Stephen C KaufmanInvestigative Ophthalmology & Visual Science April 2014, Vol.55, 511. doi: